| Assessment of Risk from Possible BSE Infectivity in Dorsal Root Ganglia |
| ─────────────────────────── |
| for |
| The Food Safety Authority of Ireland |
| ─────────────────────────── |
| DNV CONSULTING |
| ────────────────── |
| Management & Technology Solutions |
| Palace House 3 Cathedral Street London SE1 9DE United Kingdom
Det Norske Veritas Limited Org. No. 1503799 |
Business Leader, DNV Consulting |
Job No.100103
Revision 3
2nd August 2001
| Revision | Issue Date | Prepared by | Reviewed by | Approved by | Comments |
| 0 | 14th May 2001 | Philip Comer | John Spouge | Philip Comer | Draft for review |
| 1 | 24th May 2001 | Philip Comer | John Spouge | Philip Comer | Revised Draft |
| 2 | 18th July 2001 | Philip Comer | John Spouge | Philip Comer | Final Draft |
| 3 | 2nd August 2001 | Philip Comer | John Spouge | Philip Comer | Final |
The risk of exposure to the BSE infective agent due to any infectivity present in dorsal root ganglia of beef sold for domestic consumption has been assessed for the population of the Republic of Ireland for the year 2000 and the risk reduction due to removing meat from the bone estimated.
Dorsal root ganglia (DRG) are part of the peripheral nervous system and are located within the vertebral column. DRG connect to the spinal cord but would not normally be removed with the spinal cord when that is removed as SRM at the time of slaughter. Some cuts of beef, notably T-bone steaks and some cuts of rib of beef, are sold to the consumer with the vertebral column and there is therefore the possibility that people could be exposed to potentially infectious material.
In 2000 some 1.9 million cattle were slaughtered in Ireland, but of these only 205,700 (11%) were for domestic consumption. Of the cattle slaughtered for domestic consumption 5% were older than 3 years whilst the percentage of all cattle slaughtered is 30%. A statistical model of the BSE epidemic in Ireland has been used to estimate that 79 cattle were slaughtered in the calendar year before clinical onset of BSE in 2000. Of these 79 only 0.9 would have been slaughtered for the domestic market. Animals slaughtered in the calendar year before clinical onset of BSE are assumed to have a significant level of BSE infectivity, although this would be less than that in a clinical case. Animals slaughtered more than a calendar year before onset are assumed to have no significant infectivity. The model predicts that none of the animals slaughtered in the calendar year before clinical onset of BSE were less than 3 years of age.1 The model also predicts that the numbers slaughtered in the calendar year before clinical onset will reduce from 79 to 38 in 2001 and 20 in 2002. The predicted risk level would reduce in proportion to these numbers. The actual position should be monitored to check that these predictions remain valid.
The risk has been estimated using an event tree method that can be evaluated using a probabilistic risk assessment approach. This allows the range and uncertainty in the input data to be assessed. Two key assumptions in the assessment relate i) to the fraction of DRG that would remain in the meat when bone is removed from the meat in a butchers shop, and ii) to the fraction of DRG present in a bone in cut such as a T-bone steak that would be consumed. For the former two alternative assumptions are considered; in Case 1 it is assumed that 1% of the DRG are cut away with the meat, and in Case 2 that only 0.1% of the DRG are cut away with the meat. For the latter, a distribution of values with a normal distribution having a 95 percentile range from 5% to 95% has been used.
The DRG Task Force initiated a survey of abattoirs and butchers and a consumer survey to obtain information on the amount of beef sold on the bone in Ireland. The abattoir survey indicated that T-bone steaks would be produced from almost half (44%) of the animals slaughtered for domestic consumption.
The total infectivity ingested for Case 1.1 (where products such as T-bone steaks and rib of beef may be sold) is estimated to be 0.6 human oral ID50 units, with a 95 percentile range from 0.003 to 110. This infectivity would be distributed over all the beef eating population of Ireland, and the average individual risk for those that eat beef regularly (the 67% of the population who eat beef weekly) is estimated to be 2 x 10-7 human oral ID50 units per person per year, with a 95 percentile range of 1 x 10-9 to 4 x 10-5. The maximum individual risk for those eating T-bone steaks frequently (once a week) is estimated to be 7 x 10-6 human oral ID50 units per person per year, with a 95 percentile range of 4 x 10-8 to 1 x 10-3.
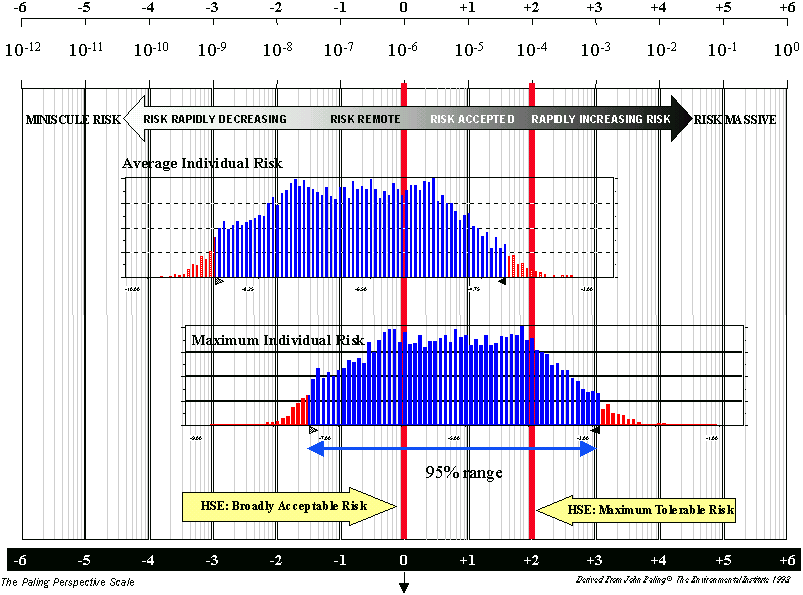
The distribution of the individual risk results is shown in Figure 1 a) on a risk perspective scale. This is a logarithmic scale and includes the risk acceptance criteria in use by the UK Health and Safety Executive for comparison. From the figure it can be seen that the median value for the average individual risk is within the level of what would normally be considered acceptable. However, the range of values extend towards the top end of the range that may be considered tolerable.
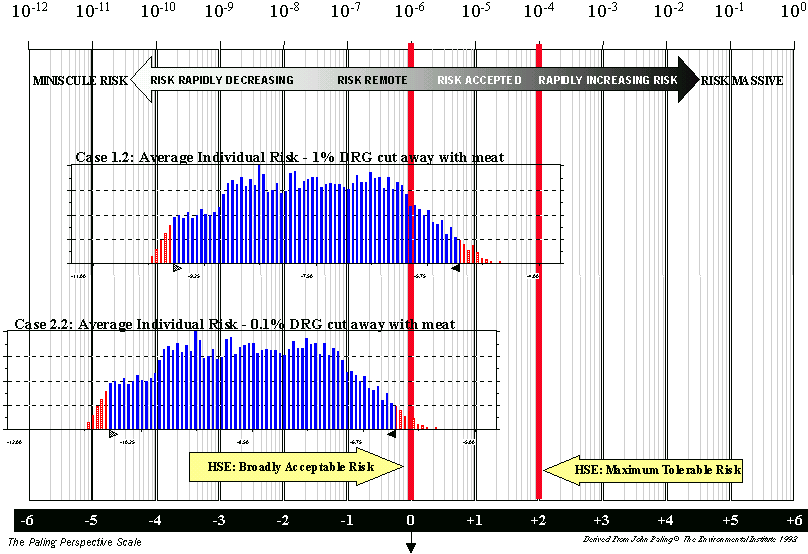
If all meat is sold off the bone, then the median value of the total infectivity ingested is estimated to be 0.08 human oral ID50 units (Case 1.2), and the median value of the individual risk is reduced to 3 x 10-8 human oral ID50 units. The distribution for this case is shown in Figure 1 b) and is now mainly below the 1 in a million risk acceptance level.
As noted above, the model of BSE epidemic in Ireland predicted that there were no animals slaughtered in the calendar year before clinical onset of BSE at less than 3 years of age. This would suggest that there is no risk associated with animals slaughtered when less than 3 years old, which represent 95% of the animals used for domestic consumption. However, in the probabilistic assessment it is assumed that there is a low probability of some animals being slaughtered in the calendar year before clinical onset. In the risk assessment, 93% of the overall risk is estimated to be due to animals slaughtered at more than 3 years old. Thus over 90% of the risk reduction could be achieved by banning the sale of beef on the bone only from cattle slaughtered at over 3 years of age, rather than from all cattle.
An alternative scenario (Case 2) has also been evaluated with a less pessimistic assumption about the amount of infectivity cut away with the meat by a butcher taking meat from the bone (0.1% as opposed to 1%). This change in assumption makes little difference when meat is sold on the bone (the total infectivity consumed being 0.5 human oral ID50 units as compared to 0.6) as most of the exposure is through the bone in meat. When meat is not sold on the bone, this change in assumption reduces the risk by a factor of 10. The total infectivity consumed reduces to 0.008 human oral ID50 units and the average individual risk reduces to 3 x 10-9 human oral ID50 units per person per year. This latter result is shown in Figure 1 b), and it can be seen that this distribution is now all below the 1 in a million “broadly acceptable” level.
An alternative approach to assessing the risk is to consider the exposure to infectivity for a person who eats a bone in cut taken from an animal with significant infectivity. The infectivity present in one DRG is estimated to range from zero to 22 human oral ID50 units, with a median value of 0.1. There is about a 50% chance that any one T-bone would contain a DRG, and 72% of all DRG (average weight 0.5 grams) would contain less than 1 human oral ID50 unit. The chance of any one T-bone steak consumed in 2000 having more than 1 human oral ID50 unit of infectivity is estimated to be 7 x 10-7 (about one in a million).
| a) | Case 1.1: Average and Maximum Individual Risk |
| b) | Cases 1.2 & 2.2: All Meat Sold Off Bone |
| ────────── | |
| 1 | In the probabilistic assessment, zero animals slaughtered in the calendar year before onset is assumed to have a 95 percentile range from 0 to 3. |
| 1. INTRODUCTION 1.1 Background 1.2 Study Objectives 1.3 Approach 2. BEEF PRODUCTION AND CONSUMPTION 2.1 Beef Production 2.2 Location of Dorsal Root Ganglia 2.3 Beef Consumption 2.4 Exposure to DRG 3. BSE IN IRELAND 3.1 Overview 3.2 Infectivity in Animals Slaughtered 3.3 Source Term 4. INFECTIVITY IN CNS TISSUE 4.1 Infectious dose for cattle 4.2 Species barrier 4.3 Infectious Dose for Humans 5. RISK ASSESSMENT 5.1 Overall Approach and Measures of Risk 5.2 Event Tree 5.3 Risk Evaluation 5.4 Results 5.4.1 Case 1 5.4.2 Case 2 5.4.3 Exposure from an infected animal 6. REFERENCES |